The clinical utility of tissue biomarkers is linked to the stain’s analytical sensitivity and consistency. To enable effective IHC quality assurance Visiopharm has developed Qualitopix:
- Trust your staining through AI-driven quantification and monitoring
- Get started easily with a frictionless set up and intuitive workflow
- Be prepared for new demands and regulations


Ensuring the accuracy and reliability of laboratory testing is paramount. This talk explores the vital role played by NordiQC (Nordic Immunohistochemical Quality Control) in enhancing the quality and standardization of diagnostic testing in pathology laboratories. I will discuss NordiQC’s objectives, operational approach, and the outcomes it has yielded. Additionally, I will introduce several valuable tools for IHC laboratories, which can be accessed on the NordiQC website.

Dr Rasmus Røge, Hematopathologist and Clinical Assoc. Prof, Aalborg University
Dr Rasmus Røge is a pathologist and Clinical Associate Professor affiliated with Aalborg University. His daily work focuses on hematopathology, specializing in the study and diagnosis of blood-related disorders. Dr Røge has been actively involved in the Nordic Immunohistochemical Quality Control (NordiQC) since 2012, an international external quality assessment scheme that serves diagnostic pathology laboratories worldwide. With more than 30 publications, his research primarily centers around quality assessment, methodology in Immunohistochemistry and introduction, validation and optimization of new biomarkers in pathology.
Standardization of immunohistochemistry (IHC) staining quality assurance (QA) is critical for diagnostic accuracy. Pathologists currently assess stain quality subjectively, comparing control sections to patient tissue. Qualitopix (Visiopharm, Denmark), a cloud-based artificial intelligence (AI) platform for IHC staining QA, uses quantitative analysis for scoring cell lines-derived, stained and digitized control slides. To establish the reliability of Qualitopix, we conducted a study to validate the precision of the digital pathology (DP) pipeline consisting of the scanners and the image analysis algorithms used.
Dirk Vossen, PhD1, Regan Fulton, MD2, PhD, Jeppe Thagaard, PhD1, Henrik Høeg, MSc1, Stine Amtoft Nielsen, MSc1, Scott Crawford, PhD2, Mateusz Tylicki, BSc (Hons)1, Andrew M Bellizzi, MD3, Sven van Kempen, Tri Q4. Nguyen, MD4, Ghlowy Gerritsen4
- Visiopharm A/S
- Array Science, LLC
- University of Iowa Hospitals & Clinics
- UMC Utrecht
 Sheila Hansen
Categories:
21255
Validating an AI-based analytic tool for IHC staining QA: precision studies of the digital pathology pipeline
Sheila Hansen
Categories:
21255
Validating an AI-based analytic tool for IHC staining QA: precision studies of the digital pathology pipeline
Standardization of immunohistochemistry (IHC) staining quality assurance (QA) is critical for diagnostic accuracy. Pathologists currently assess stain quality subjectively, comparing control sections to patient tissue. Qualitopix (Visiopharm, Denmark), a cloud-based artificial intelligence (AI) platform for IHC staining QA, uses quantitative analysis for scoring cell lines-derived, stained and digitized control slides. To establish the reliability of Qualitopix, we conducted a study to validate the precision of the digital pathology (DP) pipeline consisting of the scanners and the image analysis algorithms used.
Omar Z. Baba, MD1, Dhananjay Chitale, MD1, Kyle Perry, MD1, Nilesh Gupta, MD1, Oudai Hassan, MD1, Emily Stebens, MLS1, Kevin Daniels1, Margeaux Schmidt, MLT1, Danielle Pirain2, Henrik Høeg2, Mateusz Tylicki2, J. Mark Tuthill, MD1
- Pathology and Laboratory Medicine Department, Henry Ford Health, Detroit, Michigan, United States
- Visiopharm, Hørsholm, Denmark
 Sheila Hansen
Categories:
21254
Improving PD-L1 quality control using a dynamic rangecell line and Qualitopix analysis
Sheila Hansen
Categories:
21254
Improving PD-L1 quality control using a dynamic rangecell line and Qualitopix analysis
Immunohistochemistry (IHC) with PD-L1 is in use to predict ICI response in NSCLC patients. Inter-observer, inter- and intra-laboratory variability is, however, a known issue. In predictive IHC and to a lesser extent in panel based prognostic IHC, quality assurance principles and methods are becoming more relevant for optimal scoring. To improve scoring of PD-L1, artificial intelligence (AI) might be the way forward. Before AI can be used for daily practice, however, laboratories need to make sure that their IHC is a consistent assay instead of a routine stain.
Nils ‘t Hart1, Lars Gal1, Morten Grønning Nielsen2 and Mateusz Tylicki2
- Isala, Zwolle, The Netherlands
- Visiopharm, Hørsholm, Denmark
 Sheila Hansen
Categories:
21253
Inter-laboratory variability of HER2 low analysis using artificial intelligence
Sheila Hansen
Categories:
21253
Inter-laboratory variability of HER2 low analysis using artificial intelligence
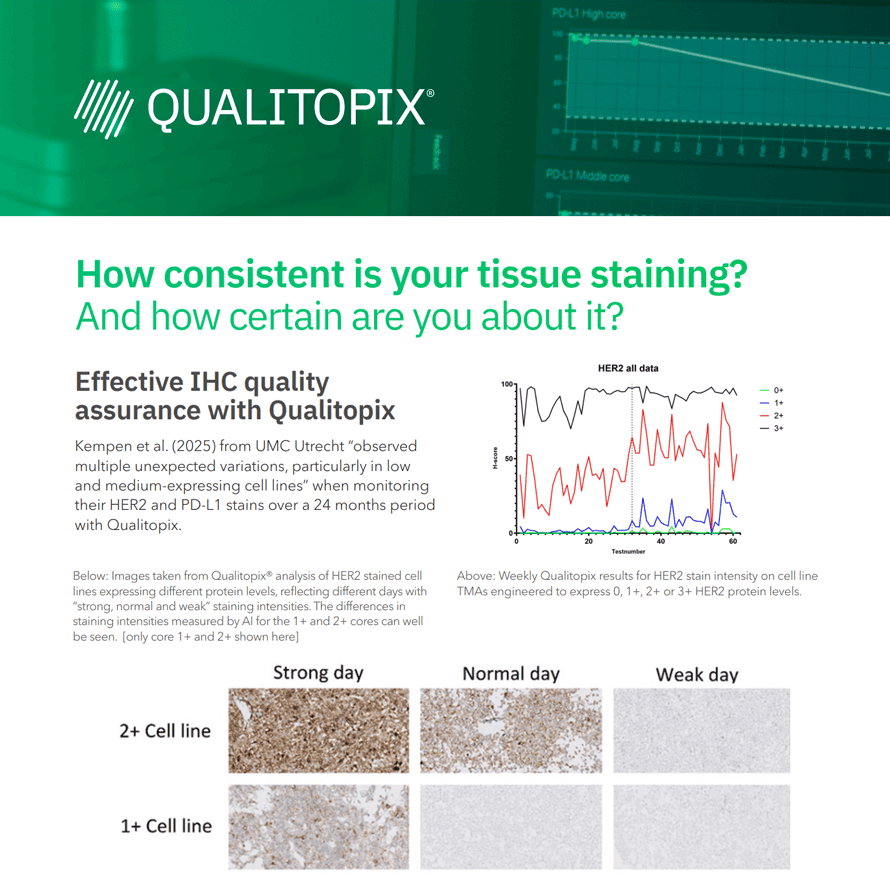
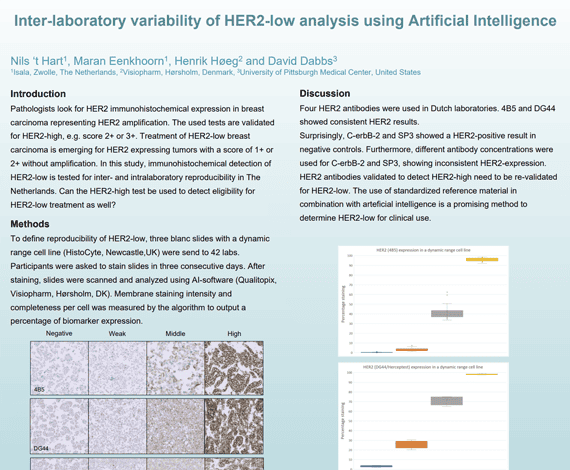
Pathologists look for HER2 immunohistochemical expression in breast carcinoma representing HER2 amplification. The used tests are validated for HER2-high, e.g. score 2+ or 3+. Treatment of HER2-low breast carcinoma is emerging for HER2 expressing tumors with a score of 1+ or 2+ without amplification. In this study, immunohistochemical detection of HER2-low is tested for inter- and intralaboratory reproducibility in The Netherlands. Can the HER2-high test be used to detect eligibility for HER2-low treatment as well?
Nils ‘t Hart1, Maran Eenkhoorn1, Henrik Høeg2 and David Dabbs3
- Isala, Zwolle, The Netherlands
- Visiopharm, Hørsholm, Denmark
- University of Pittsburgh Medical Center, United States
 Sheila Hansen
Categories:
Sheila Hansen
Categories: