

- HER2 expression is a critical biomarker in breast cancer, and HER2 status is routinely determined by IHC and/or in situ hybridization (ISH). According to the classical binary classification defined by the ASCO-CAP 2018 guidelines, tumors are categorized as HER2-positive (IHC 3+ or IHC 2+/ISH-positive) or HER2-negative (IHC 2+/ISH-negative, IHC 1+, or IHC 0)
- Accurate and timely identification of HER2 IHC status by pathologists is key for treatment selection in breast cancer. This is particularly relevant in the metastatic setting where patients with HER2-expressing tumors may be eligible for therapies such as T-DXd (a HER2-directed antibody-drug conjugate)
- Clinical benefit of T-DXd has expanded the clinical relevance of low levels of HER2 expression, with approvals based on the DESTINY-Breast04 and DESTINY-Breast06 clinical trials for patients with HER2-low (IHC 1+ or IHC 2+/ISH-negative) and HER2-ultralow (IHC 0 with faint or incomplete membrane staining in ≤10% of tumor cells) mBC
- Analyses of mBC samples originally scored as HER2 IHC 0 and IHC 1+ (HER2-negative) have showed that a substantial proportion of tumors may be reclassified as HER2-ultralow or HER2-low, and that inter-and intra-observer concordance among pathologists is variable, particularly at the lower levels of the HER2 IHC expression spectrum
- As the clinical relevance of HER2-low and HER2-ultralow has increased, accurate identification of very low HER2 expression has become increasingly important. Supporting pathologists’ confidence in interpreting these cases highlights the value for standardized protocols, additional training, and digital tools to support HER2 IHC scoring decisions
- Advances in the analysis of WSIs have transformed breast pathology by enabling workflows that support computational algorithms; CPa tools can help enhance diagnostic precision, reproducibility, and clinical decision-making
- This study builds on prior real-world findings11 of manual scoring by 3 pathologists and compares 4 standalone CPa tools versus pathologist-derived consensus scoring
Savitri Krishnamurthy, MD1, Dhanrajan Tiruchinapalli, PhD, MBA2, Clara Lam, PhD, MPH2, Simon M. Collin, PhD3, Rosemary Taylor, MSc4, Linlin Luo, MSci2, Anupriya Dutta, PhD2, Ehab El-Gabry, MD5*, Michele Sue-Ann Woo, PhD5, Grace Kwon, PharmD, MBA5, Robert Egger, PhD6, Jennifer Hipp, MD, PhD6, Lauren Brunner, Ph6, Jeppe Thagaard, PhD7, Thomas W. Ramsing, MSc7, Henrik Hoeg, MSc7, Wonkyung Jung, MD8, Heon Song, MSc8, Chang Ho Ahn, MD, PhD8, Vladimir Kravtsov, MSc9, Patrick Frey, PhD9, Ralf Banisch, PhD9, Stella Redpath, PhD2
- Department of Pathology, Division of Pathology and Laboratory Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX, USA
- Medical Affairs, AstraZeneca Pharmaceuticals LP, Gaithersburg, MD, USA
- Global Medical Affairs, AstraZeneca, Cambridge, UK
- Oncology Biometrics, AstraZeneca, Macclesfield, UK
- US Medical Affairs, Daiichi Sankyo, Inc., Basking Ridge, NJ, USA
- PathAI, Inc., Boston, MA, USA
- Computational Pathology, Visiopharm, Hørsholm, Denmark
- Lunit, Seoul, Republic of Korea
- MindPeak GmbH, Hamburg, Germany
*At the time of the study.
 Sheila Hansen
Categories:
27476
HER2 APP, breast cancer based algorithm for precise scoring of HER2-null, ultra-low, and low expression as an alternative to ASCO/CAP visual scoring: an analysis of 422 cases
Sheila Hansen
Categories:
27476
HER2 APP, breast cancer based algorithm for precise scoring of HER2-null, ultra-low, and low expression as an alternative to ASCO/CAP visual scoring: an analysis of 422 cases
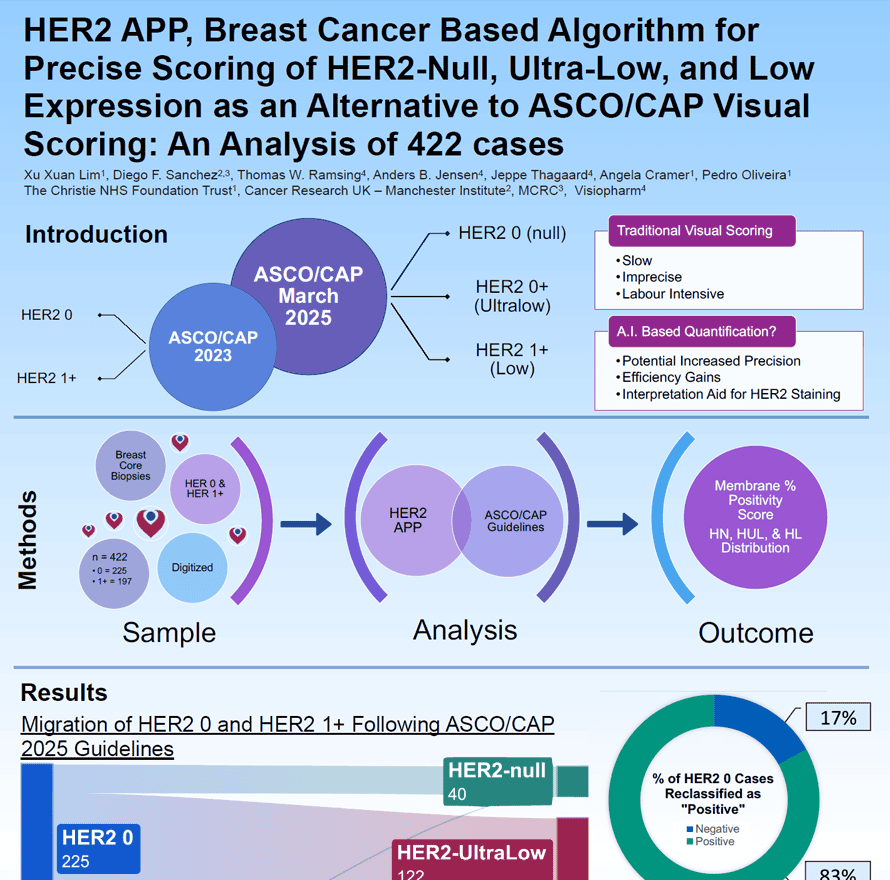

Accurate HER2 assessment in breast cancer is essential for guiding targeted therapy, yet traditional visual scoring is challenging. The updated ASCO/CAP reporting template adds HER2-null, ultra-low, and low categories, increasing interpretation complexity. This study evaluates an AI-based algorithm (HER2 APP) for precise, efficient quantification of HER2 membrane positivity in 422 digitized biopsies.
Xu Xuan Lim1, Diego F. Sanchez2,3, Thomas W. Ramsing4, Anders B. Jensen4, Jeppe Thagaard4, Angela Cramer1, Pedro Oliveira1
- The Christie NHS Foundation Trust
- Cancer Research UK – Manchester Institute
- MCRC
- Visiopharm
Manual biomarker scoring can be both time-consuming and inconsistent — challenges that become even more critical with classifications like HER2-low and -ultralow. The Insight platform enables AI-driven, fully automated image analysis APPs to provide precise, reliable biomarker scoring. With Insight, you can eliminate variability and focus on what truly matters: making confident, informed decisions.
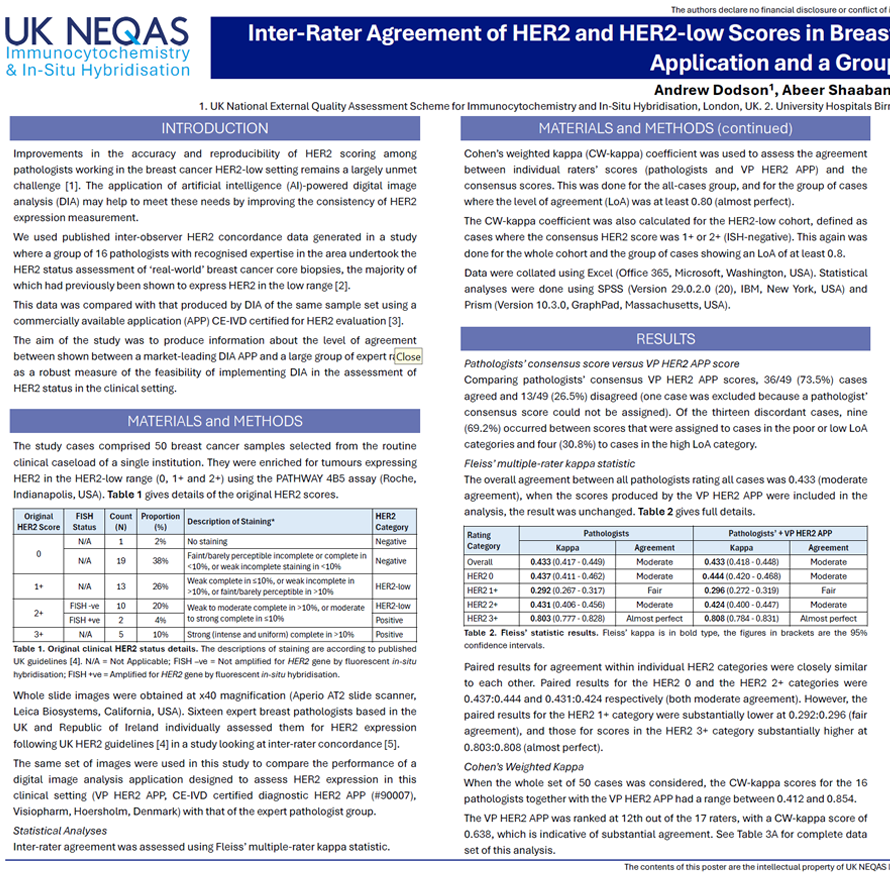

Improvements in the accuracy and reproducibility of HER2 scoring among pathologists working in the breast cancer HER2-low setting remains a largely unmet challenge. The application of artificial intelligence (AI)-powered digital image analysis (DIA) may help to meet these needs by improving the consistency of HER2 expression measurement.We used published inter-observer HER2 concordance data generated in a study where a group of 16 pathologists with recognised expertise in the area undertook the HER2 status assessment of ‘real-world’ breast cancer core biopsies, the majority of which had previously been shown to express HER2 in the low range.This data was compared with that produced by DIA of the same sample set using a commercially available application (APP) for HER2 evaluation. The aim of the study was to produce information about the level of agreement between shown between a market-leading DIA APP and a large group of expert raters as a robust measure of the feasibility of implementing DIA in the assessment of HER2 status in the clinical setting.
Andrew Dodson1, Abeer Shaaban2, Lila A Zabaglo1, Suzanne Parry1
- UK National External Quality Assessment Scheme for Immunocytochemistry and In-Situ Hybridisation, London UK.
- University Hospitals Birmingham NHS Foundation Trust Queen Elizabeth Hospital, Birmingham, UK.


- Accurate assessment of lower ranges of HER2 immunohistochemical (IHC) expression has become important since trastuzumab deruxtecan (T-DXd) was approved for treatment of patients with HER2-low metastatic breast cancer (BC).
- Findings from DESTINY-Breast06 Phase 3 trial showed that T-DXd offers a favourable outcome over standard chemotherapy in patients with HER2-low and HER2-ultra-low metastatic BC.
- Several studies showed low concordance among pathologists in distinguishing low levels of HER2 IHC expression.
- There is an emerging need for a standardized and reproducible HER2-low testing methodology.
- A dedicated EQA programme for the assessment of IHC stain quality of HER2-low testing was established by UK NEQAS ICC & ISH in 2023 providing support to clinical laboratories and gathering evidence about the reproducibility of HER2-low testing.
- We report results from the application of digital image analysis (DIA) for the HER2-low IHC assessment with a view of implementing DIA in the HER2-low EQA programme.
Lila Zabaglo1, Suzanne Parry1, Dawn Wilkinson1, Andrew Dodson1
- UK National External Quality Assessment Scheme for Immunocytochemistry and In-Situ Hybridisation, London UK.
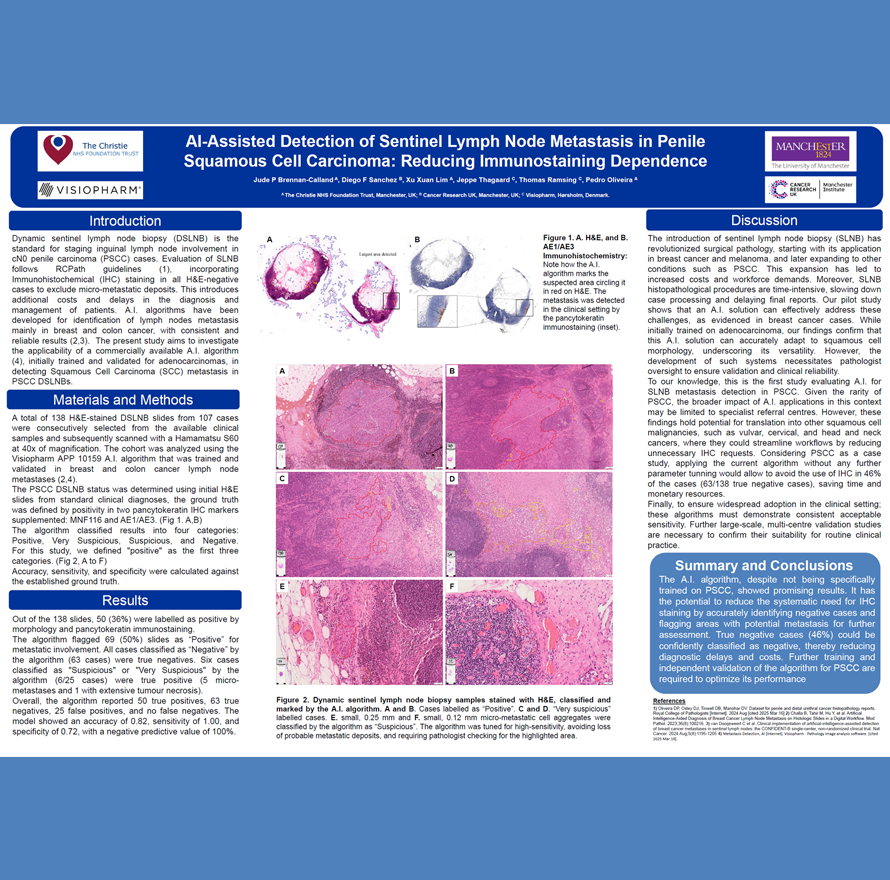

The growth in cancer immunotherapy agents requires an understanding of the immune contexture of the tumor microenvironment (TME). This can be aided by high-plex imaging and analysis to obtain phenotypes of specific cells and study their biodistribution and interactions. Imaging Mass Cytometry (IMC) is the method of choice for single-step staining and high-plex imaging of tissues, avoiding the complications of autofluorescence and cyclic imaging.
IMC has expanded its capabilities with three distinct imaging modes: Preview, Cell, and Tissue. The Preview Mode is a rapid scanning system that captures a comprehensive overview of the stained tissue, mapping out the distribution of over 40 markers and revealing tissue heterogeneity. This enables researchers to make informed decisions about which areas warrant closer examination on the same. Building on this, Cell Mode offers high-resolution imaging for detailed analysis of the Regions of Interest (ROIs) identified during Preview, all using the same slide. Tissue Mode complements these by providing a fast acquisition of the entire tissue at a lower resolution, which is optimal for quantitative pixel-based analysis of tissue biology. These modes support automated, continuous imaging of more than 40 large tissue samples (400 mm2) weekly. Following Preview Mode, the selection of ROIs for high-resolution imaging is a critical step, enhanced by automated AI algorithms to ensure it is informed by biomarker expression.
Jude P Brennan-CallandA, Diego F SanchezB, Xu Xuan LimA, Jeppe ThagaardC, Thomas RamsingC, Pedro OliveiraA
- The Christie NHS Foundation Trust, Manchester, UK
- Cancer Research UK, Manchester, UK
- Visiopharm, Hørsholm, DenmarkA/S

