
Accelerating precision in the era of targeted therapy
1. The structural shift in oncology
Oncology drug development is undergoing a fundamental structural transformation. The rapid expansion of targeted therapies, such as antibody-drug conjugates (ADCs) and bispecific antibodies, has changed how treatment eligibility is determined. Increasingly, access to therapy depends on identifying subtle quantitative differences in biomarker expression in tissues.
Weak expression levels, heterogeneous tumor distribution, and sub-cellular localization patterns are no longer academic nuances. They now directly determine whether a patient qualifies for clinically meaningful therapy. In this new landscape, precision has moved from being descriptive to being decisive.
In our previous blog, The Precision Gap in Therapy-Linked Biomarker Testing: A Structured Analysis of Diagnostic Error and Its Clinical Consequences, we discussed how Clinical Value Leakage arises when Analytical Signal Integrity and Interpretation Variability compromise this precision. These error sources are measurable and increasingly consequential as more therapies become biomarker-defined.
Technical solutions now exist to address both signal integrity and interpretation variability. AI-assisted quantification for staining quality monitoring and decision support are no longer theoretical capabilities. However, the adoption speed of these digital and AI-enabled tools remains slow. This creates a disconnect between what oncology requires and what routine anatomical pathology workflows consistently deliver. We refer to this widening delta as the Precision Gap.
2. Clinical Value Leakage: Where precision is lost
The concept of Clinical Value Leakage describes how otherwise avoidable variability in biomarker testing can compromise therapy alignment. There are two dominant contributors to this leakage: Analytical Signal Integrity and Interpretation Variability.
Signal integrity reflects variability in staining quality, pre-analytical handling, and assay performance that can distort the biological signal. Interpretation variability reflects reader-dependent differences that arise when pathologists assess complex expression patterns.
As therapy eligibility increasingly depends on marginal quantitative distinctions, even small shifts in signal or interpretation can alter treatment access for patients who’s biomarkers are near cut-off. These are not unsolvable problems, as technical solutions exist to reduce both forms of variability. Improving interpretation without stabilizing the signal leaves variability upstream, while improving signal without supporting interpretation leaves variability downstream.
The challenge today is systematic deployment.
3. The prevailing deployment model: Infrastructure-first
Over the past decade, the dominant vision for the digital pathology field has centered on large-scale enterprise transformation. This Infrastructure-First model aims to digitize the entire laboratory environment through high-throughput scanners, enterprise image- and workflow management systems (IMS/PACS), and full integration with laboratory and medical records (LIMS/HIS).
The objectives of this model are compelling. It seeks to achieve efficiency at scale and precision across all diagnostic workflows. In fully digital environments, this model enables remote collaboration, operational efficiency, and broad access to advanced analytics.
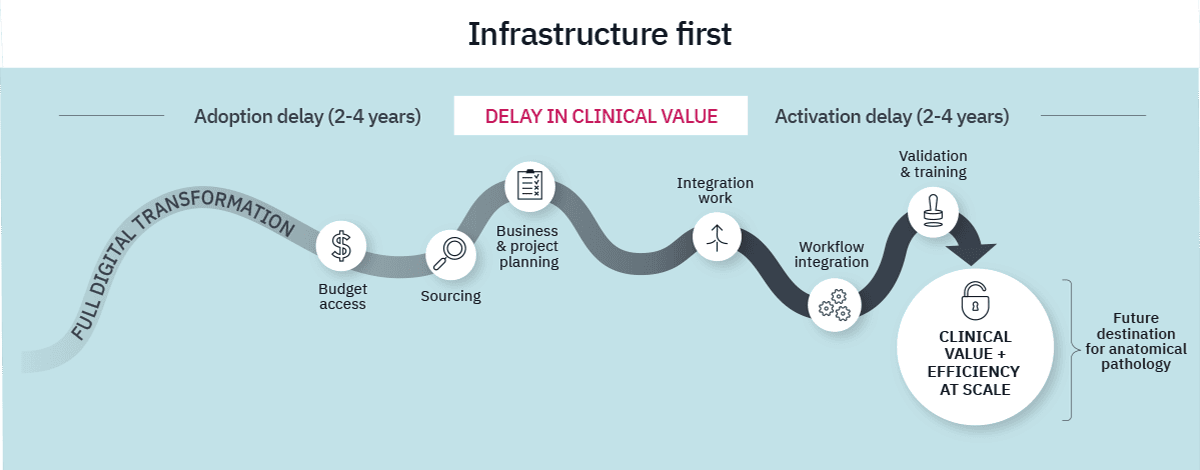
However, enterprise deployment is inherently complex and requires significant time, investment and change management. Institutions often face an initial adoption phase driven by capital access, multi-stakeholder alignment, and hardware procurement, often lasting two to four years. Only after this phase does activation begin, requiring additional years for integration, interoperability testing, validation, and organizational change management.
As a result, clinical precision is frequently deferred until the broader infrastructure is complete, often spanning four to eight years in total. In many institutions, digital transformation must compete with other strategic priorities, even as therapeutic demand from oncology departments intensifies. This creates a structural challenge where oncology innovation accelerates while precision activation remains gated by infrastructure complexity, widening the Precision Gap.
4. Therapy cannot wait for infrastructure
Based on this analysis, we propose an alternative deployment model that addresses Clinical Value Leakage without first requiring full digital transformation.
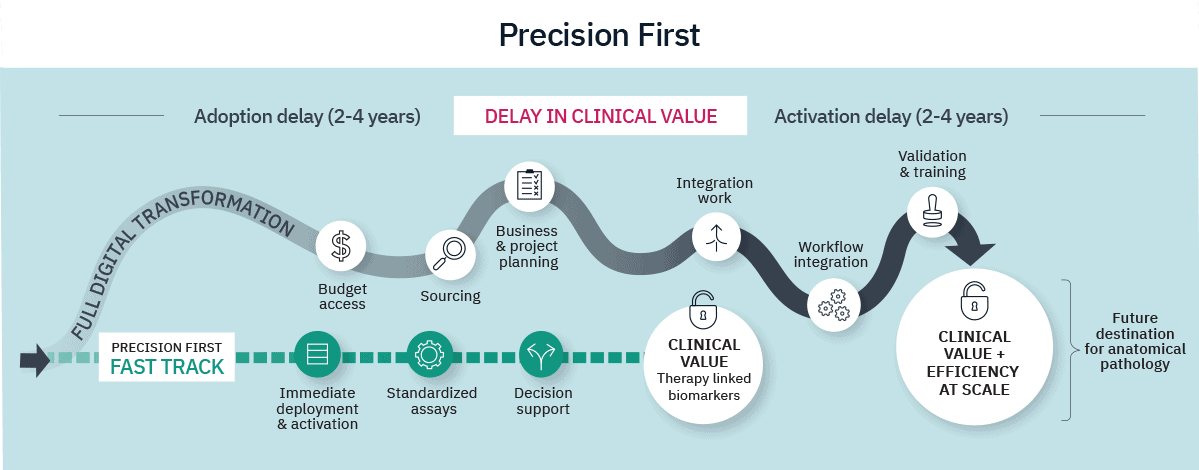
If the Infrastructure-First model begins with systems, a Precision-First model begins with the patient. This deployment model starts where clinical risk is highest: with the therapy-linked biomarkers that directly determine treatment eligibility.
These biomarkers are often lower in volume but high in biological complexity. Instead of requiring digitization of the entire laboratory before enabling precision, the Precision-First approach concentrates on addressing the two dominant contributors to Clinical Value Leakage.
4.1 AI-driven precision pathology:
Two capabilities form the technical foundation of this approach:
- Biomarker interpretation support: AI-supported quantification reduces reader-dependent variability in borderline cases. Quantitative decision support brings reproducibility to assessments that sit at therapeutic thresholds. Beyond reproducibility, AI-based scoring can achieve systematically higher sensitivity in low-expression biomarkers.
- Stain quality management: AI-based monitoring of staining performance stabilizes analytical signal integrity. By leveraging standardized control materials, staining performance can be monitored with a level of consistency previously unavailable in routine workflows.
The highest levels of precision are achieved when signal integrity and interpretation variability are aligned. Precision-First enables institutions to address one or both domains in a targeted manner, depending on their starting point and priorities.
4.2 Pragmatic deployment:
Because therapy-linked biomarkers represent focused, high-impact use cases, deployment can remain targeted and pragmatic. Precision-First does not require full enterprise digitization or extensive multi-year digital transformation to begin delivering value. It can operate with low-volume scanners and lightweight / featherweight cloud-based processing.
This approach allows clinical value to be realized incrementally. Capital exposure is limited, organizational disruption is reduced, and executive risk is distributed. Most importantly, precision is activated where the therapeutic consequences are greatest.
The oncology drug pipeline will continue to increase the precision required in biomarker testing. Low expression, spatial heterogeneity, quantitative thresholds and continuous scoring will only grow in clinical importance. Precision is therefore no longer optional; it is foundational.
Full digital transformation remains the necessary destination for anatomical pathology. Enterprise-scale digitization is essential to enable long-term efficiency and precision across all diagnostic applications.
5. Precision first, transformation always
However, full digital transformation often spans multiple years. For therapy-linked biomarkers that determine immediate treatment eligibility, critical decisions cannot wait for that timeline. Precision-First is not an alternative to enterprise transformation; it is a catalyst for it. Early deployment generates measurable clinical and operational evidence, builds internal familiarity with digital workflows, and creates institutional advocates for broader adoption. By activating precision early, institutions can reduce Clinical Value Leakage immediately while building the momentum, evidence base, and organizational confidence needed for the broader digital journey.

Michael Grunkin, PhD, CEO of Visiopharm



