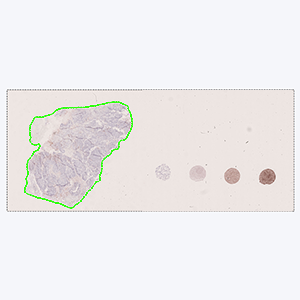
Tissue detection
Detection of sample tissue and exclusion of control tissues.
#90183
Immunotherapies directed at programmed death ligand 1 (PD-L1) and its receptor (PD-1) have enhanced the survival rate of a group of patients with advanced lung cancer. The expression of PD-L1 protein can predict which patients are more inclined to benefit from immunotherapy.
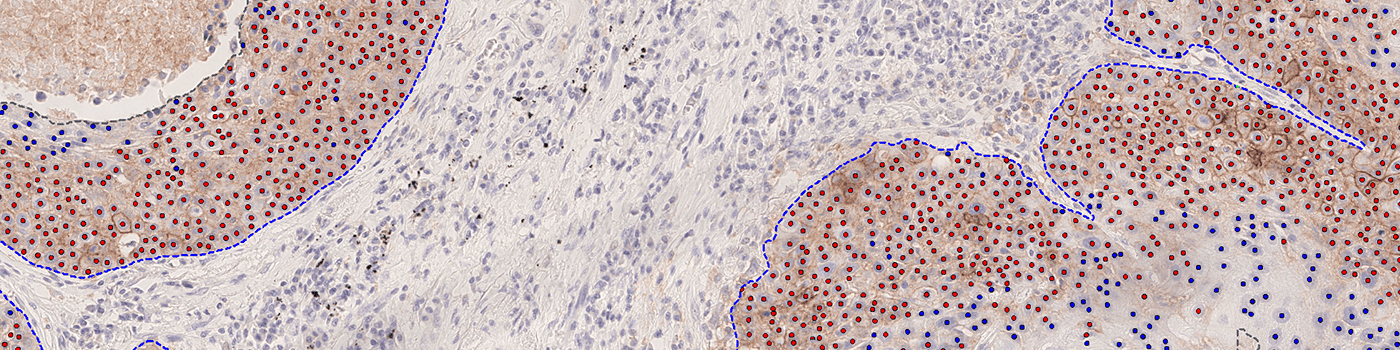
Our PD-L1 solution is fully automated and does not require manual input during analysis. The solution consists of four automated analysis steps. Once started, it automatically separates the patient tissue from control tissues and robustly identifies invasive cancer areas in the sample. Tumor cells are counted based on their PD-L1 expression and the resulting Tumor Proportion Score (TPS) for the whole tumor area is calculated.
In EU/UK: CE IVD – for use in diagnostic procedures
Quantitative Output variables
The output variables obtained from this protocol are:
Total Tumor Nuclei (#): The number of tumor nuclei within all ROIs
TPS (%): The PD-L1 Tumor Proportion Score (TPS)
Workflow
Step 1:
After scanning, the PACS/LIS/IMS informs the Visiopharm software about the new image and the analysis of the image is automatically started.
Alternatively, the analysis can be started manually by opening the image and starting the analysis.
Step 2:
The pathologist reviews the fully analyzed image. If needed, adjustments can be made. Once reviewed, pathologists sign off the case in the system.